Peer Reviewed
May-Thurner Syndrome Presenting as Recurrent Isolated Left-sided Deep Vein Thrombosis
AFFILIATIONS:
1Internal Medicine resident, Saint Peters University Hospital, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ
2Medical Student, Rutgers University, Robert Wood Johnson Medical School, New Brunswick, NJ
3Department of Internal Medicine, Saint Peters University Hospital, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ
CITATION:
Thota G, Ballam Nagaraj N, Weerasinghe K, Gribkova Y, Yegneswaran B. May-Thurner syndrome presenting as recurrent isolated left-sided deep vein thrombosis. Consultant. 2022;62(12):e7. doi:10.25270/con.2022.07.000017
Received February 13, 2022. Accepted May 11, 2022. Published online July 26, 2022.
DISCLOSURES:
The authors report no relevant financial relationships.
DISCLAIMER:
No written consent has been obtained from the patients as there is no patient identifiable data included in this case report.
CORRESPONDENCE:
Geethika Thota, MD, Saint Peter’s University Hospital, 254 Easton Ave., New Brunswick, NJ, 08901 (prathima.thota95@gmail.com)
Abstract
We present a case of a 62-year-old man who had no symptoms of venous insufficiency, with left leg swelling and pain a few days after being treated for floating deep vein thrombosis (DVT), whose condition was subsequently diagnosed as May-Thurner syndrome (MTS). This report highlights the importance of a broad differential diagnosis that includes MTS and considers anatomic abnormalities in situations of unprovoked, persistent DVT of the lower extremities.
Key words: leg pain, leg swelling, thrombosis, anticoagulation, deep vein thrombosis, recurrent
Case Presentation
A 62-year-old man with a medical history of rheumatoid arthritis and gastroesophageal reflux disease presented to the hospital with persistent left leg pain and swelling for one day. He did not report associated chest pain, shortness of breath, myalgia, fever, or chills, nor did he have associated leg numbness, tingling, or altered sensation.
He had no recent sick contacts, trauma, or travel. He had no family history of clotting disorders. A few days prior to presentation, the patient reported having similar symptoms and was found to have a floating deep vein thrombosis (DVT) in the left common femoral and greater saphenous veins for which he was started on outpatient anticoagulation.
His medications at that time included methotrexate, folic acid, pantoprazole, and apixaban. He had a history of left leg DVT 6 years ago provoked by a total knee replacement surgery and had been taking rivaroxaban for a few weeks after the event.
Physical examination. At presentation, the patient had a temperature of 36.6 °C, a heart rate of 76 beats/min, a respiratory rate of 18 breaths/min, a blood pressure of 128/78 mm Hg, an oxygen saturation level of 96% on room air, and a body mass index of 28 kg/m2.
On physical examination, the patient appeared to be in no acute distress. His abdomen was soft, nondistended, and nontender. He had significant tenderness of the left leg from ankle to thigh. His left calf measured 50.8 cm, and his right calf measured 45.7 cm. Pulses were palpable in bilateral lower extremities.
Diagnostic testing. Results of the initial laboratory tests, including complete blood cell count, comprehensive metabolic panel, and coagulation profile (prothrombin time, international normalized ratio, partial thromboplastin time), were within normal limits. A computed tomography (CT) venogram showed nonthrombotic occlusion and marked atresia of the left common iliac vein, confirming the diagnosis of nonthrombotic May-Thurner syndrome (MTS) (Figures 1 and 2).
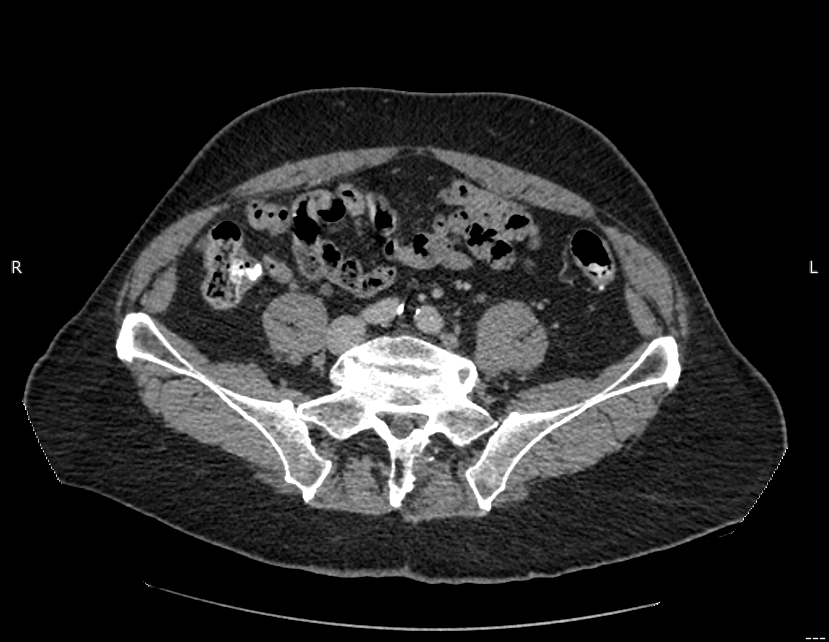
Figure 1. CT scan showing chronic occlusion of the left common iliac vein secondary to compression by the right common iliac artery.
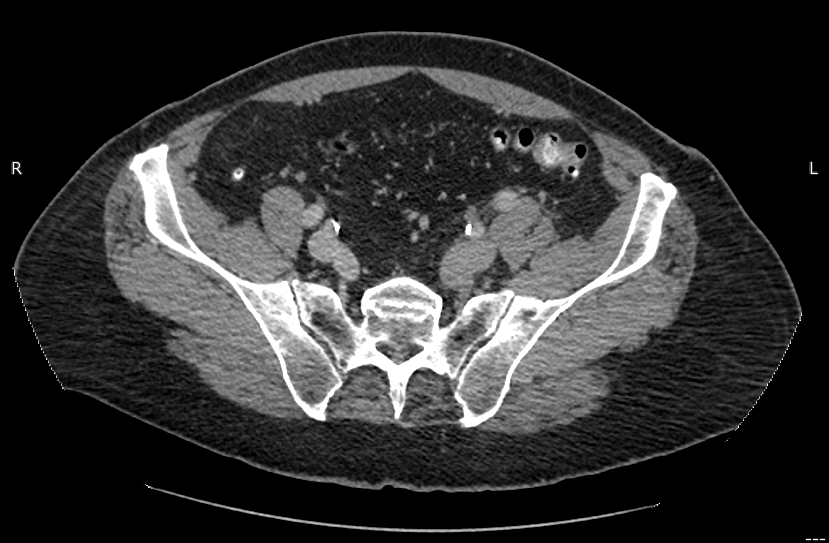
Figure 2. CT scan showing reconstitution of an enlarged left external iliac vein from the patent left internal iliac vein.
Discussion. MTS is a venous syndrome described as the compression of the left iliofemoral vein by the overlying right common iliac artery and the underlying lumbar vertebrae.1 The continued irritation of the venous endothelium by compression and pulsation of the iliac artery causes the development of intraluminal venous spurs and promotes clot formation.2 MTS is thought to account for 2% to 5% of all DVTs; however, studies of postmortem autopsies suggest that the prevalence of MTS may be much higher.3 MTS tends to occur more frequently in young women, and patients are often asymptomatic. However, when symptoms are present, patients are likely to have either acute or recurrent proximal DVT in the left lower limb or a clinical presentation of chronic venous insufficiency.1
MTS is an anatomical abnormality that leads to outflow obstruction of the iliofemoral vein by external compression.4,5 This syndrome is highly variable in location and level of obstruction, with the most common form being the compression of the left iliac vein by the overlying right common iliac artery and the fifth lumbar vertebrae.2 While many patients remain asymptomatic, there are identifiable risk factors for MTS, including female sex, oral contraceptive use, scoliosis, radiation exposure, and hypercoagulable disorders.3,6-8 The most common presenting symptoms are lower extremity swelling and claudication. Symptoms of chronic venous insufficiency, such as lower extremity edema, skin discoloration or ulceration, and female pelvic congestion syndrome, may also be seen.2,6,7
When MTS is suspected, a color Doppler ultrasonography scan is usually performed initially as it is noninvasive and less expensive than other diagnostic tools.9,10 However, the Doppler has a relatively low sensitivity for visualization of vein compression and cannot ascertain the location of the obstruction or the number of spurs in the lumen.1,9 The modalities of choice in the suspected cases are CT venography, magnetic resonance venography, intravenous ultrasonography, and conventional venography.9,10 However, despite multiple imaging options, the gold standard for diagnosis of MTS is conventional venography with intravascular ultrasonography.9,10 This form of imaging allows for information about luminal diameter and can reveal the level of obstruction in the blood vessel in addition to having some therapeutic benefits.
Differential diagnoses to consider include the more common causes of iliac vein compression: malignancy, lymphadenopathy, hematoma, and cellulitis. Additionally, various etiologies of uterine enlargement, aortoiliac aneurysms, retroperitoneal fibrosis, and osteophytes should remain potential diagnoses until MTS is confirmed.2 A coagulative disorder workup and a malignancy screening should be performed prior to the diagnosis of MTS to rule out other etiologies.2 MTS is more likely if the patient is young, female, uses an oral contraceptive, and has lower limb involvement.1 However, in this case, the patient was an older man who had extensive MTS for several years but did not develop a thrombus or any symptoms until age 62 years, indicating that MTS should not be overlooked as a diagnosis for any patient population.
Treatment for MTS is not needed unless symptomatic. In the past, open surgical procedures were performed; with the latest advancement in technology, less invasive interventional radiology-guided repairs are more popular. Clot removal with mechanical thrombectomy, pharmacologic thrombolysis, and stenting of the anatomic defect is the mainstay of treatment.6,9 For patients in whom thrombolysis is contraindicated, a multitude of various endovascular thrombectomies, such as rheolytic, rotational, or suction, may be used instead.6,9,10 Although the use of a prophylactic inferior vena cava filter is usually not recommended, initial anticoagulation for patients cannot be delayed, as it is associated with a higher risk of pulmonary embolism.3,11 Low molecular weight heparin or fondaparinux is preferred over unfractionated heparin because of decreased risk for bleeding and heparin-induced thrombocytopenia.2 Rivaroxaban has also been shown to be safe in patients with iliofemoral vein thrombosis.12 There is no standardized guideline for anticoagulation therapy for patients with MTS, and sources vary with regard to duration of antiplatelet therapy. Some studies suggest antiplatelet therapy for 6 weeks to 3 months combined with aspirin for an indefinite amount of time, while others advise antiplatelet therapy for at least 6 months to prevent restenosis and aspirin indefinitely.1,13 Expert opinion from a hematologist can be helpful in developing an anticoagulation plan that considers specific risks and benefits for each individual patient.
After intervention, imaging with duplex ultrasonography can be conducted to confirm the patency of the iliac vein. Subsequently, patients should be screened at 6 months postdiagnosis and then annually with duplex ultrasonography. If patients develop symptoms or if there is a defect detected on duplex ultrasonography, cross-sectional imaging is necessary.13
Patient outcome. On the day of presentation, the patient underwent interventional radiology-guided venography; however, the chronically occluded vessel could not be crossed for stenting at that time. Thrombolysis of the clot was achieved with alteplase infusion and subsequent heparin for 3 days. A repeat interventional radiology-guided venography was performed to stent the persistent left common iliac vein stricture. The patient was not started on aspirin because he was allergic but was started with clopidogrel for at least 6 weeks. The patient tolerated the procedure well and was discharged home on anticoagulation therapy with apixaban, 5 mg twice daily. The patient remained asymptomatic on outpatient follow-up.
Conclusion. Our case highlights the importance of considering anatomic abnormalities in situations of unprovoked, persistent DVT of the lower extremities and urges health care providers to broaden their differential diagnoses and diagnostics to include anatomical abnormalities that could be causing the patient’s symptomatology.
1. Poyyamoli S, Mehta P, Cherian M, et al. May-Thurner syndrome. Cardiovasc Diagn Ther. 2021;11(5):1104-1111. https://doi.org/10.21037/cdt.2020.03.07
2. Mangla A, Hamad H. May-Thurner syndrome. StatPearls. StatPearls Publishing. Updated May 14, 2022.
3. Harbin MM, Lutsey PL. May-Thurner syndrome: History of understanding and need for defining population prevalence. J Thromb Haemost. 2020;18(3):534-542. https://doi.org/10.1111/jth.14707
4. Farina R, Foti PV, Iannace FA, et al. May Thurner syndrome: description of a case with unusual clinical onset. J Ultrasound. 2020;25(2):309-313. https://doi.org/10.1007/s40477-020-00497-0
5. Peters M, Syed RK, Katz M, et al. May-Thurner syndrome: a not so uncommon cause of a common condition. Proc (Bayl Univ Med Cent). 2012;25(3):231-233. https://doi.org/10.1080/08998280.2012.11928834
6. Kalu S, Shah P, Natarajan A, Nwankwo N, Mustafa U, Hussain N. May-thurner syndrome: a case report and review of the literature. Case Rep Vasc Med. 2013;2013:740182. https://doi.org/10.1155/2013/74018
7. Antignani PL, Lazarashvili Z, Monedero JL, et al. Diagnosis and treatment of pelvic congestion syndrome: UIP consensus document. Int Angiol. 2019;38(4):265-283. https://doi.org/10.23736/S0392-9590.19.04237-8
8. Tewoldemedhin B, Tewoldemedhin NK, Ahmed S, Karki S, Micheal M. A curious presentation of May-Thurner syndrome with isolated iliofemoral deep vein thrombosis. Cureus. 2022;14(2):e22124. doi:10.7759/cureus.22124
9. Radaideh Q, Patel NM, Shammas NW. Iliac vein compression: epidemiology, diagnosis and treatment. Vasc Health Risk Manag. 2019;15:115-122. https://doi.org/10.2147/VHRM.S203349
10. Knuttinen MG, Naidu S, Oklu R, et al. May-Thurner: diagnosis and endovascular management. Cardiovasc Diagn Ther. 2017;7(suppl 3):S159-S164. https://doi.org/10.21037/cdt.2017.10.14
11. Kaltenmeier CT, Erben Y, Indes J, et al. Systematic review of May-Thurner syndrome with emphasis on gender differences. J Vasc Surg Venous Lymphat Disord. 2018;6(3):399-407.e4. https://doi.org/10.1016/j.jvsv.2017.11.006
12. Kang JM, Park KH, Ahn S, et al. Rivaroxaban after thrombolysis in acute iliofemoral venous thrombosis: a randomized, open-labeled, multicenter trial. Sci Rep. 2019;9(1):20356. https://doi.org/10.1038/s41598-019-56887-w
13. Fereydooni A, Stern JR. Contemporary treatment of May-Thurner syndrome. J Cardiovasc Surg (Torino). 2021;62(5):447-455. https://doi.org/10.23736/S0021-9509.21.11889-0


